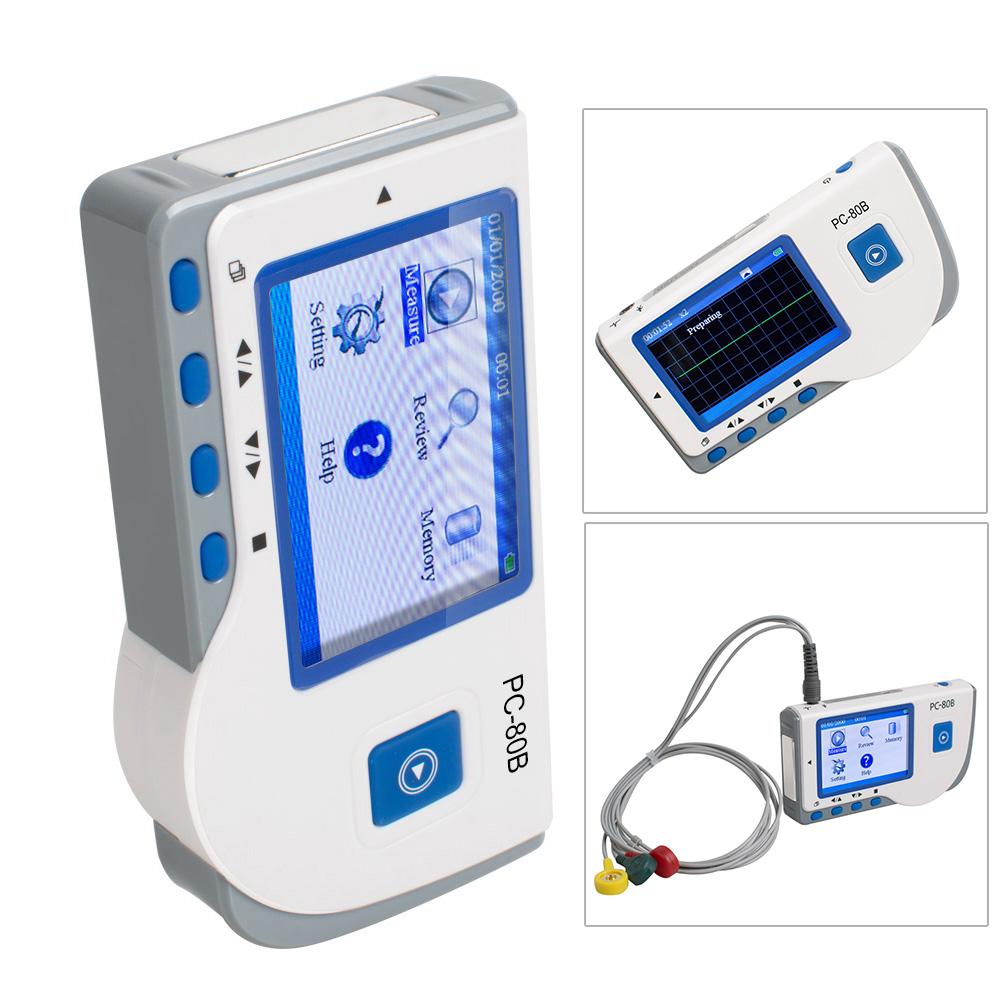
Description:
PC -80B Easy ECG Monitor is intended formeasuring and recording the ECG signal and average heart rate
of adult patient. It is applicable for use in clinics and homes,and convenient to operate by the patients
themselves. It can be used for continuous measuring with the adhesive ECG pads and lead wire connected to the device.
Specification:
1.Display
Display Mode: Color LCD display
Screen Size (mm): 40*57.6
2.ECG Measurement
The number of measuring channel: Single channel ECG signal
Electrodes: 3 embedded metal electrodes or 3 adhesive ECG pads connected to the connector of
the device with lead wire
ECG Bandwidth: 1Hz-40Hz
Internal Noise Level: ≤30uVp-p
Heart Rate Range: 30-240bpm
Heart Rate Accuracy: ±2bpm or ±2%, whichever is greater
Display Scale: 5mm/mV ±10%
Common-mode rejection ratio (CMRR): ≥60dB
Wave Form Sweeping Speed: 20mm/s±10%
Measurement Modes: the detecting ECG signal can be detected by the following lead selection:
1) Palm (both hands on unit)
2) Chest (right hand and chest)
3) Leg (right hand and left leg)
4) ECG lead wire
3.Power
Power Supply: 2*AAA alkaline batteries
Supply voltage range: 2.7-3.3VDC
Max average working current: ≤50mA
Auto power-off: 35s±5s (without operation)
4.Classification
The type of protection against electric shock: Internally powered equipment
The degree of protection against electric shock: Type B appiled parts
The degree of protection against harmful ingress of liquid: Ordinary equipment without protecting
against ingress of liquid.
Electro-magnetic Compatibility: Group I, Class B
5.Data Memory
Up to 10-hour data records can be saved in the built-in memory
Stored data records can be uploaded to the computer by data cable
6. Operating environment:
Operating Temperature: 5°C-40°C
Operating Humidity: 30%-80%
Atmospheric pressure: 70kPa-106kPa
Packing List:
1 x Machine main unit (without battery)
1 x English user's manual
1 x Data cable
1 x ECG lead wire
FDA
declaration :
Statement:The sale of this item
may be subject to regulation by the U.S. Food and Drug Administration and state
and local regulatory agencies. If so, do not bid on this item unless you are an
authorized purchaser. If the item is subject to FDA regulation, I will verify
your status as an authorized purchaser of this item before shipping of the
item.(CICI-China-86-18328503374)
This
item has been cleaned and treated according to the manufacturer's instructions.
The
Fingertip Pulse Oximeter is certified with the US FDA 510K No. K070371, the CE
& TUV of Eureope and it is on the Australian Register of Therapeutic Goods
(ARTG) with the code 136606.
The
Powered Surgical Instrument / Speed 808 System is certified with the US FDA
510(k) Number:K132989
The
Powered Surgical Instrument / Hair Remove Device is certified with the US FDA
510(k) Number:K180353
The
Powered Surgical Instrument / Hair Remove System is certified with the US FDA
510(k) Number:K141973
massager,
vacuum, light induced heating / Slimming Treatment Device is certified with the
US FDA 510(k) Number:K161892