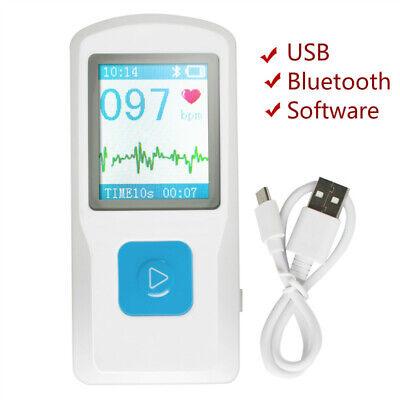
Features:
1.77 color TFT-LCD for quick and easy ECG checking with a simple finger touch- Convenient operation and immediate accurate results- Built-in rechargeable lithium battery enables continuous recording of up to 500 cases on a full charge- Bluetooth transmission for seamless data transfer- ECG data can be securely saved to the CLOUD Platform for permanent storage and convenient analysis- Historical data can be easily printed whenever needed for convenient diagnosis
Packing List:
Package list: 1×Main Machine 1×USB Line
Description:
Introduction
PM10 Portable ECG Monitor is a device for checking ECG,which is applicable for family and
individual user, it is a good helper to early prevent from cardiovascular diseases and reduce risks.
Intelligent design, achieves remote health management by using with mobile application, it can
automatically start measurement, store ECG, upload data, download health conclusion and obtain
doctor advice at the first moment.
Physical Characteristic:
Battery Voltage:DC3.7V
Operating Current:≦150mA
Dimension: 100mm(L)×45mm(W)×15mm(H)
Weight:about 60g
Package list:
1×Main Machine
1×USB Line
FDA Declaration:
Statement:The sale of this item may be subject to regulation by the U.S. Food and Drug Administration and state and local regulatory agencies. If so, do not bid on this item unless you are an authorized purchaser.If the item is subject to FDA regulation, we will verify your status as an authorized purchaser of this item before shipping of the item.(Seller Name: Sara;Country: China;City: Beijing;TEL.:86-15210656055).
This item has been cleaned and treated according to the manufacturer's instructions.
The Powered Surgical Instrument / Speed 808 System is certified with the US FDA 510(k) Number:K132989
The Powered Surgical Instrument / Hair Remove Device is certified with the US FDA 510(k) Number:K180353
The Powered Surgical Instrument / Hair Remove System is certified with the US FDA 510(k) Number:K141973
massager, vacuum, light induced heating / Slimming Treatment Device is certified with the US FDA 510(k) Number:K161892
Ultrasound ,Ultrasonic Treatment Device is certified with the US FDA 510(k) Number:K161892
1.US stock items shipping service: UPS 3-5 BUSINESS DAYS (weekend and festival not included) from our warehouse in the US, only to United-States. China stock items will be shipped by epacket or Airmail post within 1 business days.
1.If you receive a defective item which you want to return, please contact us within 2 days from the day you receive the item.
2.All return items must be returned with its original packaging and accessories. If we confirm the damage happened during the shipment, we will bear the shipping cost for return.
3.We will refund the money to you when we get the return items. Or replace item for you free of charge.